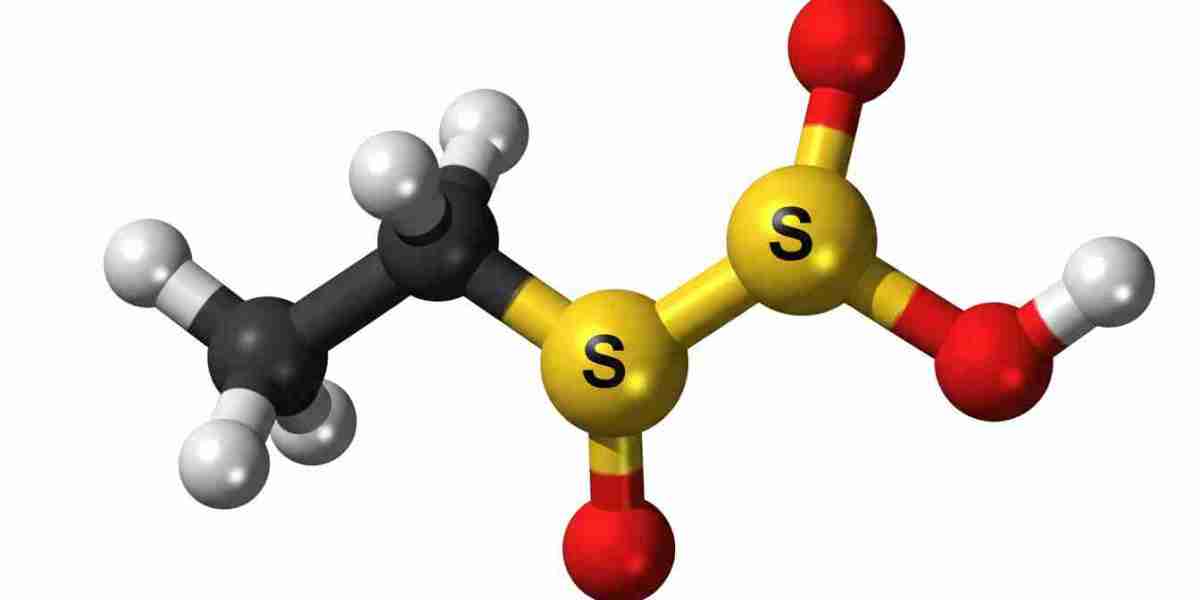
Ethylene sulfate is a chemical compound that belongs to the group of organosulfur compounds. It is widely known for its role as an intermediate in chemical synthesis and industrial applications. Due to its reactive nature, ethylene sulfate is used in various processes, especially in the production of other chemicals and materials. Understanding its structure, behavior, and uses is essential for industries that rely on chemical manufacturing.Ethylene sulfate is not commonly found in everyday consumer products, but it plays a crucial role behind the scenes in laboratories and industrial environments. Its importance lies in its ability to participate in chemical reactions that lead to the formation of more complex compounds.
Chemical Structure and Properties
Ethylene sulfate has a cyclic structure that includes sulfur and oxygen atoms along with carbon atoms. This structure makes it highly reactive compared to many other compounds. It appears as a colorless to pale yellow liquid under standard conditions.One of the key properties of ethylene sulfate is its high reactivity with nucleophiles. This means it can easily react with substances that donate electrons, making it useful in chemical synthesis. It also has moderate solubility in water and is more soluble in organic solvents.Its boiling point, density, and stability depend on environmental conditions such as temperature and pressure. However, it is generally considered unstable when exposed to moisture or heat for extended periods.
Production of Ethylene Sulfate
Ethylene sulfate is typically produced through the reaction of ethylene oxide with sulfuric acid or related sulfur-containing compounds. This process requires controlled conditions to ensure safety and efficiency.The production involves careful temperature regulation because the reaction can be highly exothermic. Industrial setups use specialized equipment to handle the chemical safely and to prevent unwanted side reactions.In laboratory settings, smaller quantities can be synthesized for research purposes. However, due to its hazardous nature, strict safety protocols are followed during its preparation.
Industrial Applications
Ethylene sulfate is mainly used as an intermediate in the production of other chemicals. It plays a significant role in manufacturing polymers, resins, and certain pharmaceutical compounds.In the plastics industry, it is sometimes used to modify materials or to introduce specific chemical groups into polymer chains. This helps improve the properties of the final product, such as strength, flexibility, or resistance to heat.Additionally, ethylene sulfate is used in chemical research for studying reaction mechanisms and developing new compounds. Its reactivity makes it a valuable tool for chemists working on advanced materials.
Role in Chemical Synthesis
One of the most important uses of ethylene sulfate is in chemical synthesis. It acts as an alkylating agent, meaning it can transfer an ethylene group to other molecules. This property is essential in creating complex organic compounds.Chemists often use ethylene sulfate to introduce functional groups into molecules, which can then be further modified. This step is crucial in the development of pharmaceuticals, agrochemicals, and specialty chemicals.Because of its efficiency in reactions, ethylene sulfate helps reduce the number of steps required in synthesis, making processes more cost-effective and time-saving.
Health Hazards and Risks
Ethylene sulfate is considered a hazardous substance due to its toxic and potentially carcinogenic properties. Exposure can occur through inhalation, skin contact, or ingestion, each posing serious health risks.Short-term exposure may cause irritation to the eyes, skin, and respiratory system. Symptoms can include redness, burning sensation, coughing, and difficulty breathing. Prolonged exposure may lead to more severe health issues, including damage to internal organs.There is also concern about its potential to cause genetic mutations, which is why it is handled with extreme caution in both industrial and laboratory environments.
Safety Precautions and Handling
Proper safety measures are essential when working with ethylene sulfate. Workers must wear protective equipment such as gloves, goggles, and lab coats to minimize exposure.It should be handled in well-ventilated areas or under a fume hood to prevent inhalation of vapors. Storage conditions should be cool and dry, away from incompatible substances such as strong bases or oxidizing agents.Emergency procedures should be in place in case of accidental exposure or spills. This includes having access to safety showers, eye wash stations, and proper disposal methods for contaminated materials.
Environmental Impact
Ethylene sulfate can have negative effects on the environment if not managed properly. It may contaminate water sources and soil, posing risks to aquatic life and ecosystems.Due to its reactivity, it can break down into other harmful substances that persist in the environment. Therefore, industries must follow strict regulations for disposal and waste management.Efforts are being made to develop safer alternatives and greener processes that reduce the environmental footprint of chemicals like ethylene sulfate.
Regulations and Guidelines
Many countries have established regulations to control the use and handling of ethylene sulfate. These guidelines are designed to protect workers, consumers, and the environment.Organizations such as occupational safety agencies provide limits for exposure and recommend best practices for handling hazardous chemicals. Companies must comply with these regulations to ensure safe operations.Proper labeling, documentation, and training are also required to maintain safety standards in workplaces where ethylene sulfate is used.
Future Prospects and Research
Research on ethylene sulfate continues to explore its potential applications and safer handling methods. Scientists are working to understand its behavior better and to develop alternatives with lower toxicity.Innovations in green chemistry aim to reduce reliance on hazardous substances by creating environmentally friendly compounds. This could lead to reduced use of ethylene sulfate in the future.At the same time, its effectiveness in chemical synthesis ensures that it will remain an important compound in research and industry for years to come.
Conclusion
Ethylene sulfate is a highly reactive and important chemical compound used in various industrial and research applications. While it offers significant benefits in chemical synthesis, it also poses serious health and environmental risks.Understanding its properties, uses, and safety measures is essential for anyone working with or studying this compound. With ongoing research and strict regulations, efforts are being made to minimize its risks while maximizing its benefits in modern chemistry.